A multicenter, randomized, placebo-controlled, double-blind phase 3 trial with open-arm comparison indicates safety and efficacy of nephroprotective therapy with ramipril in children with Alport's syndrome - ScienceDirect
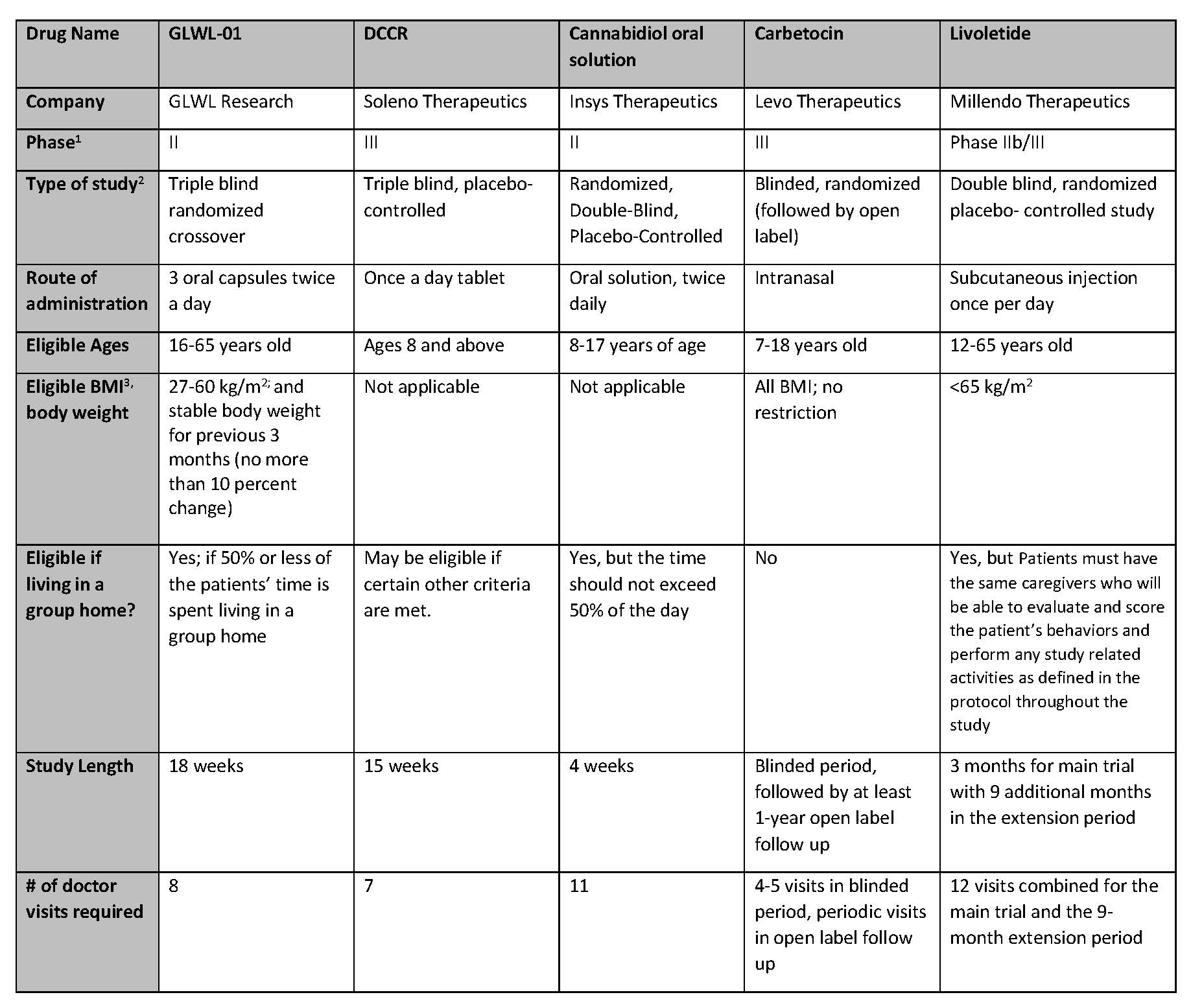
Summary Of Active Clinical Trials For Prader-Willi Syndrome Hyperphagia - Prader-Willi Syndrome Association | USA
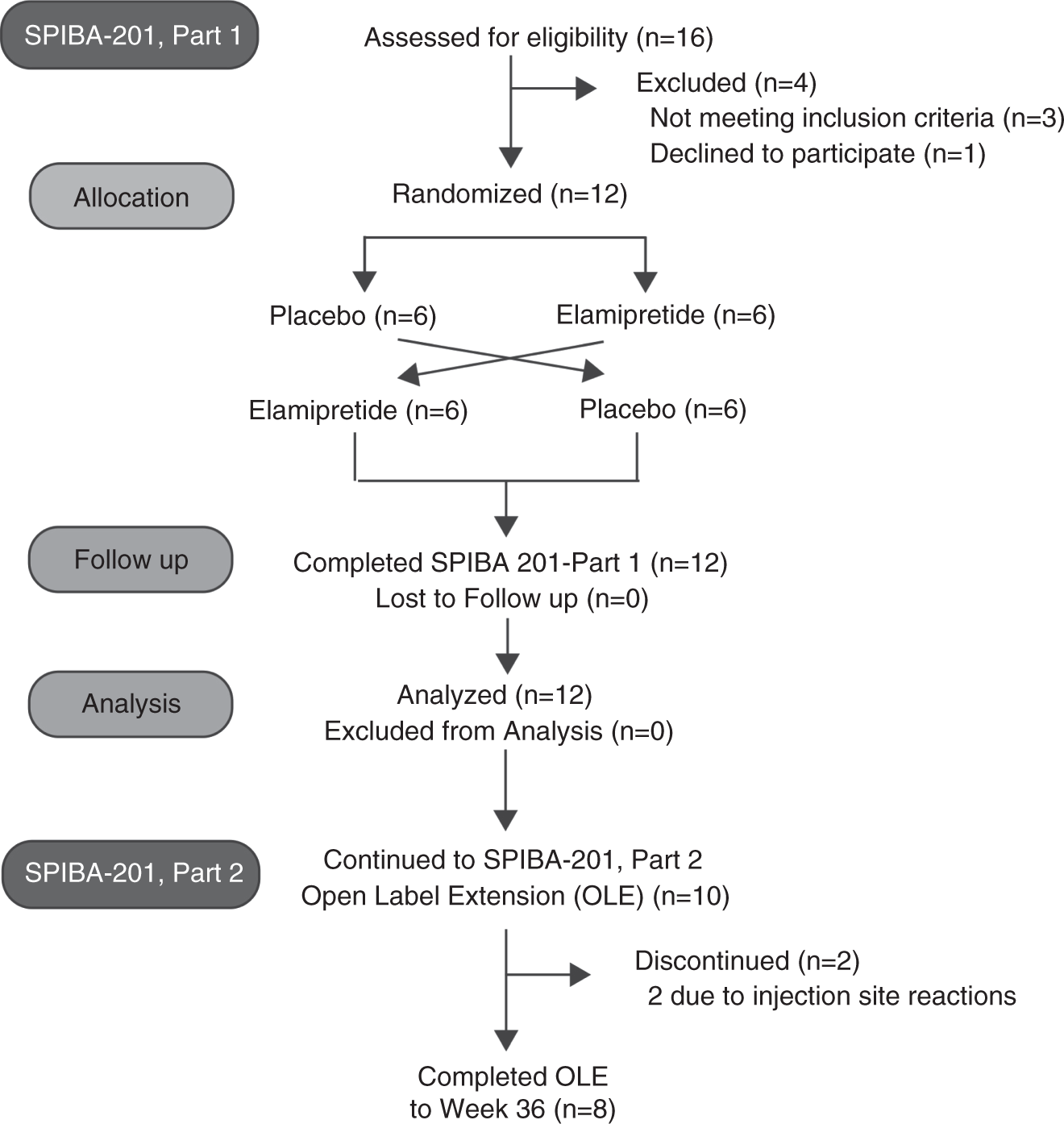
A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome, a genetic disorder of mitochondrial cardiolipin metabolism | Genetics in Medicine

Comparing the effects of ipragliflozin versus metformin on visceral fat reduction and metabolic dysfunction in Japanese patients with type 2 diabetes treated with sitagliptin: A prospective, multicentre, open‐label, blinded‐endpoint, randomized ...

PDF) GI-REASONS: a novel 6-month, prospective, randomized, open-label, blinded end point (PROBE) trial

Bioavailability of everolimus administered as a single 5 mg tablet versus five 1 mg tablets: a randomized, open-label, two-way crossover study of healthy volunteers

Clopidogrel plus aspirin versus aspirin alone for reducing embolisation in patients with acute symptomatic cerebral or carotid artery stenosis (CLAIR study): a randomised, open-label, blinded-endpoint trial - The Lancet Neurology
![PDF] GI-REASONS: A Novel 6-Month, Prospective, Randomized, Open-Label, Blinded Endpoint (PROBE) Trial | Semantic Scholar PDF] GI-REASONS: A Novel 6-Month, Prospective, Randomized, Open-Label, Blinded Endpoint (PROBE) Trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a0653b7b38e796366a33a888639ab58e6f15b68e/4-Figure1-1.png)
PDF] GI-REASONS: A Novel 6-Month, Prospective, Randomized, Open-Label, Blinded Endpoint (PROBE) Trial | Semantic Scholar

Alteplase versus tenecteplase for thrombolysis after ischaemic stroke (ATTEST): a phase 2, randomised, open-label, blinded endpoint study - The Lancet Neurology
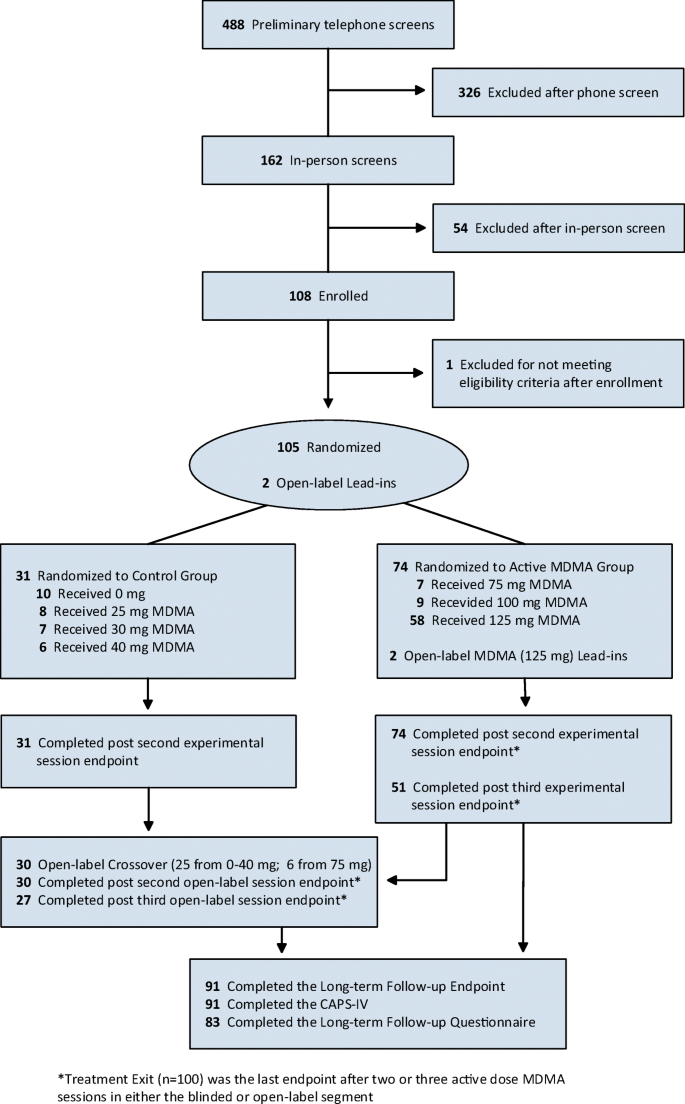
Long-term follow-up outcomes of MDMA-assisted psychotherapy for treatment of PTSD: a longitudinal pooled analysis of six phase 2 trials | SpringerLink

Frontiers | Randomized, Placebo-Controlled, Double-Blind and Open-Label Studies in the Treatment and Prevention of Acute Diarrhea With Enterococcus faecium SF68 | Medicine
PLOS Medicine: Preventing microalbuminuria with benazepril, valsartan, and benazepril–valsartan combination therapy in diabetic patients with high-normal albuminuria: A prospective, randomized, open-label, blinded endpoint (PROBE) study

Methodology of a large prospective, randomised, open, blinded endpoint streamlined safety study of celecoxib versus traditional non-steroidal anti-inflammatory drugs in patients with osteoarthritis or rheumatoid arthritis: protocol of the standard care ...

1 SECOND AUSTRALIAN NATIONAL BLOOD PRESSURE STUDY (ANBP-2) Enalapril/ACEI vs. HCTZ, n = 6,083 Randomized, open-label (blinded endpoint review) All CV events. - ppt download

A Prospective, Randomized, Open-Label, Blinded, Endpoint Study Exploring Platelet Response to Half-Dose Prasugrel and Ticagrelor in Patients with the Acute Coronary Syndrome: HOPE-TAILOR Study | Semantic Scholar

PDF) Alteplase versus tenecteplase for thrombolysis after ischaemic stroke (ATTEST): a phase 2, randomised, open-label, blinded endpoint study | Dheeraj Kalladka - Academia.edu
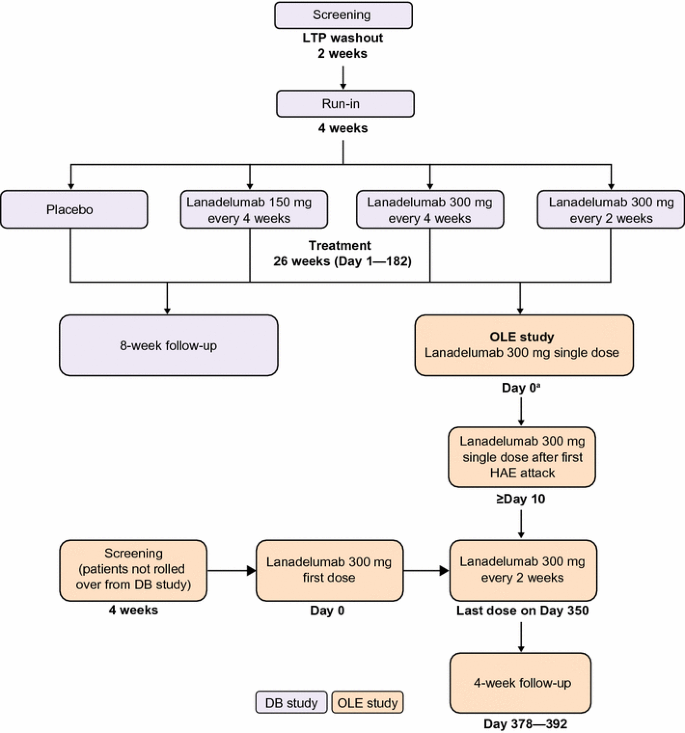
An open-label study to evaluate the long-term safety and efficacy of lanadelumab for prevention of attacks in hereditary angioedema: design of the HELP study extension | Clinical and Translational Allergy | Full