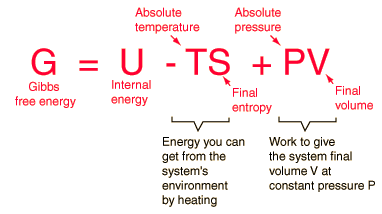
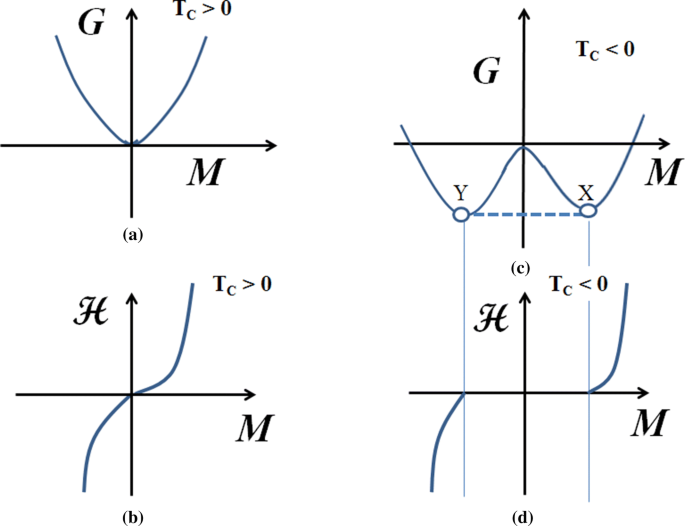
MathType on Twitter: "The #Helmholtz free energy is associated with the work done by a closed system at constant volume and temperature. Given a partition function, this energy is an efficient way

CSIR-UGC NET - Internal energy , Helmholtz free energy and enthalpy ( in hindi ) Offered by Unacademy
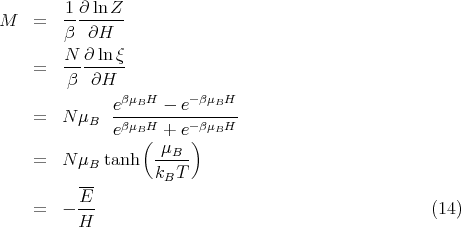
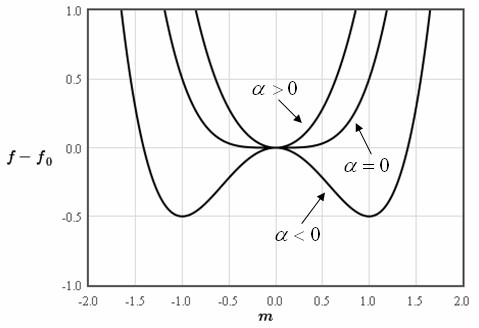
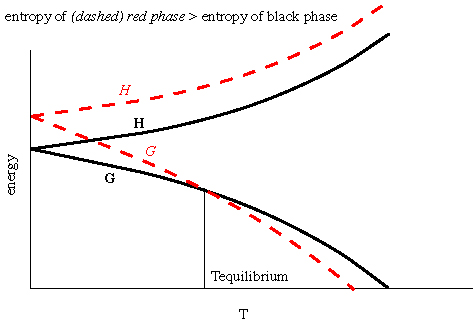
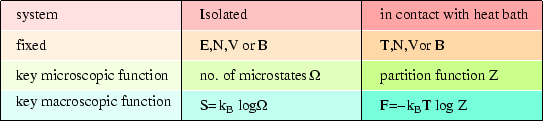
LECTURE 10 Simple Applications of Statistical Mechanics We have seen that if we can calculate the partition function ∑ Z = e-βEr r (1) then we can derive just about anything we want to know from the partition function such as the mean internal energy, the ...

Direct calculation of the solid-liquid Gibbs free energy difference in a single equilibrium simulation: The Journal of Chemical Physics: Vol 139, No 10