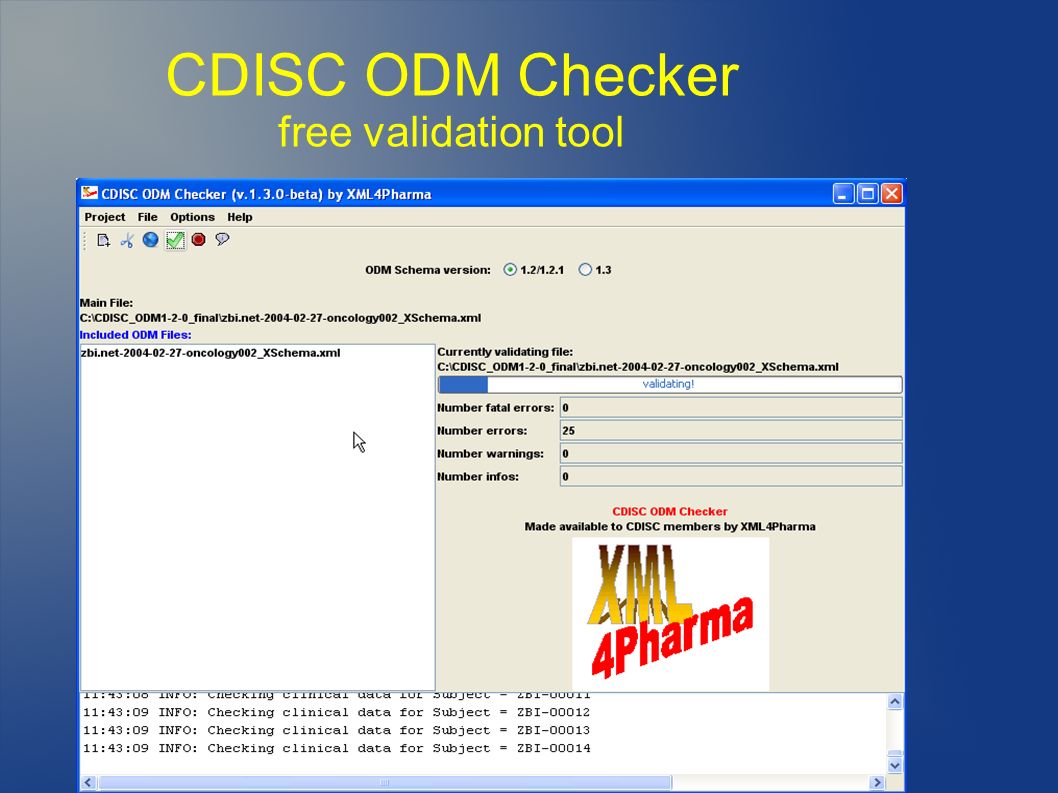
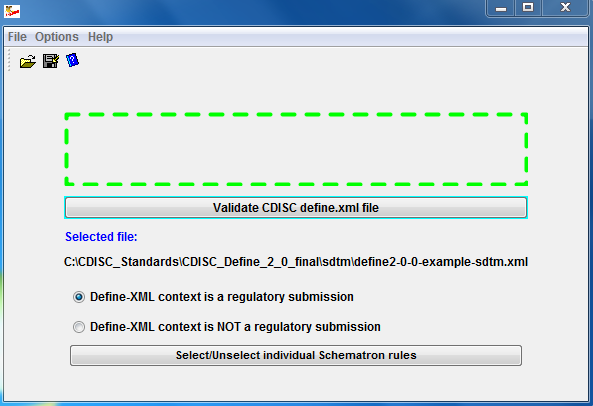
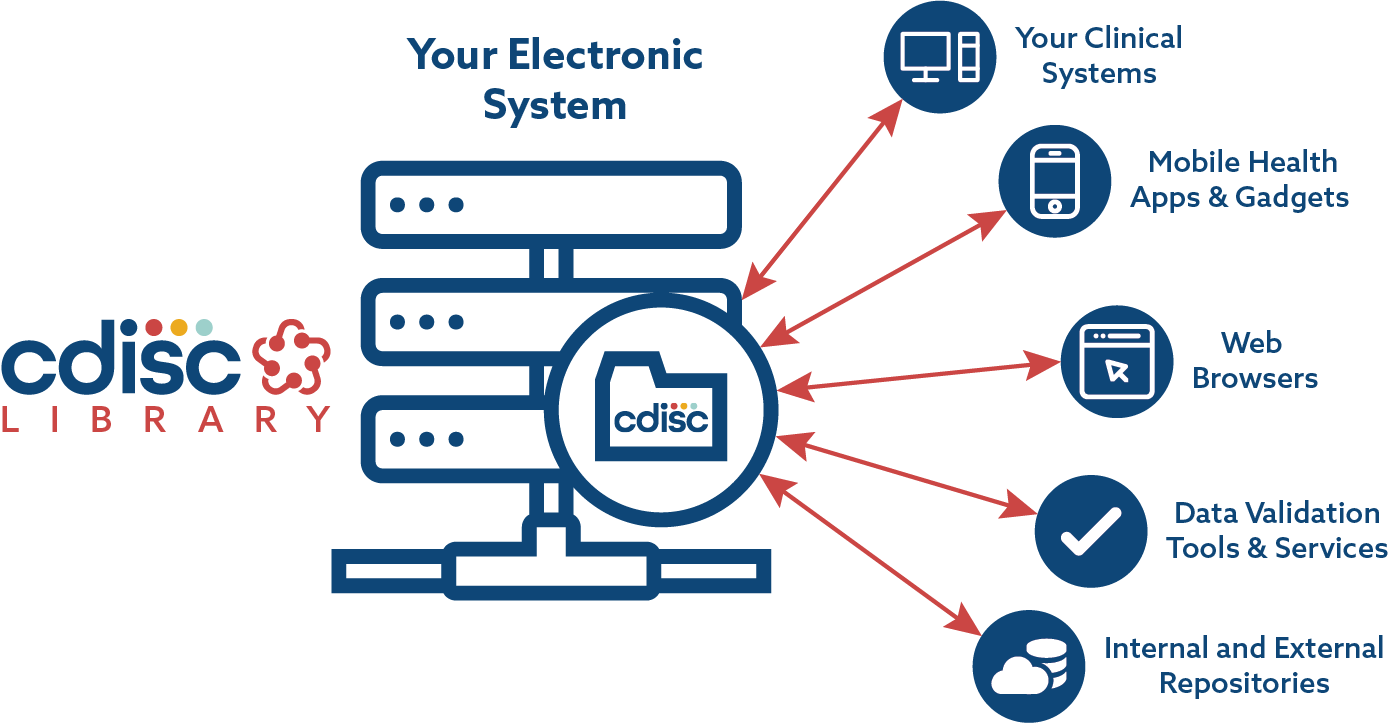
PLOS ONE: ODM Data Analysis—A tool for the automatic validation, monitoring and generation of generic descriptive statistics of patient data

A standard-driven approach for electronic submission to pharmaceutical regulatory authorities - ScienceDirect
![PDF] In-Depth Review of Validation Tools to Check Compliance of CDISC SDTM-Ready Clinical Datasets | Semantic Scholar PDF] In-Depth Review of Validation Tools to Check Compliance of CDISC SDTM-Ready Clinical Datasets | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4ea1ea0e99952efdcc712efee14e0d18e3b4208e/4-Figure3-1.png)
PDF] In-Depth Review of Validation Tools to Check Compliance of CDISC SDTM-Ready Clinical Datasets | Semantic Scholar
![PDF] In-Depth Review of Validation Tools to Check Compliance of CDISC SDTM-Ready Clinical Datasets | Semantic Scholar PDF] In-Depth Review of Validation Tools to Check Compliance of CDISC SDTM-Ready Clinical Datasets | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4ea1ea0e99952efdcc712efee14e0d18e3b4208e/3-Figure2-1.png)
PDF] In-Depth Review of Validation Tools to Check Compliance of CDISC SDTM-Ready Clinical Datasets | Semantic Scholar