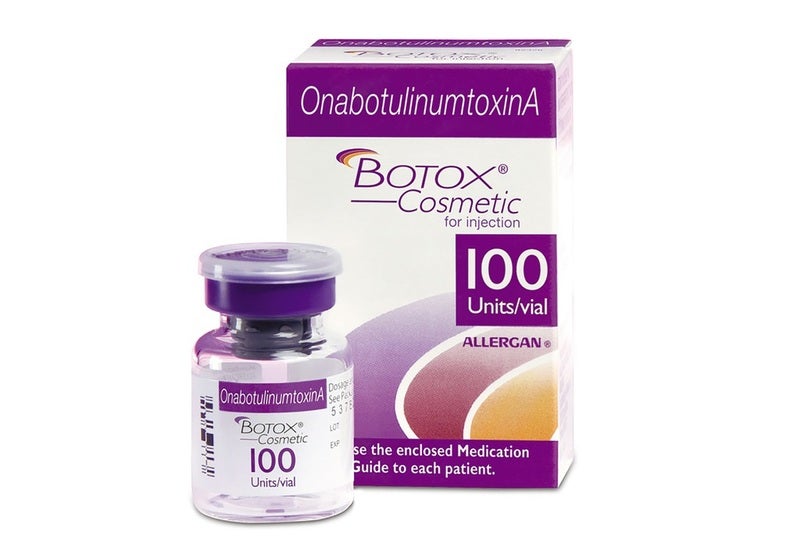
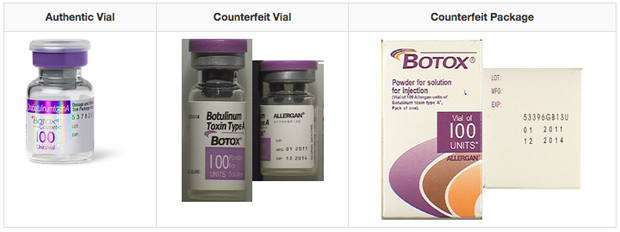
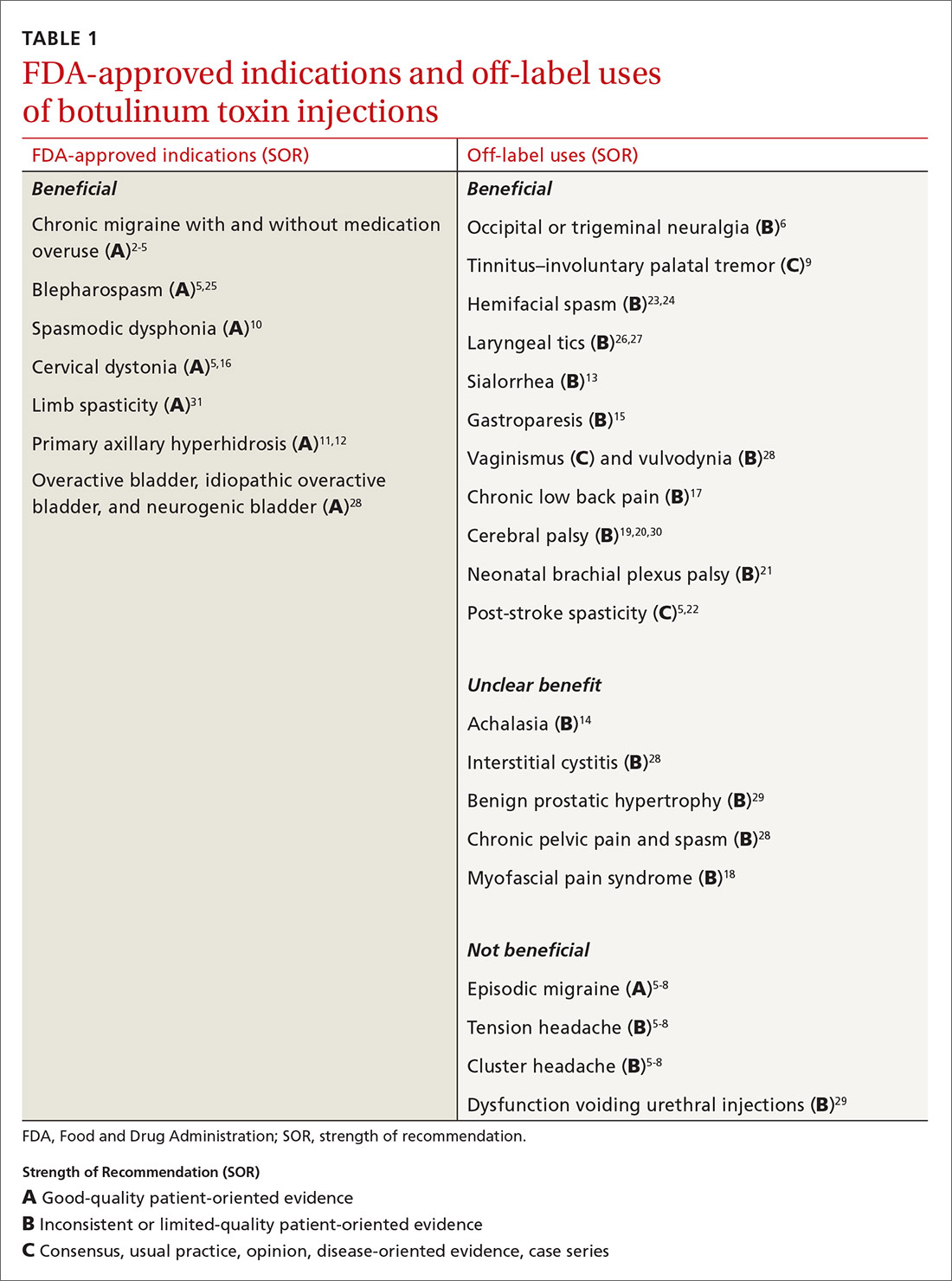
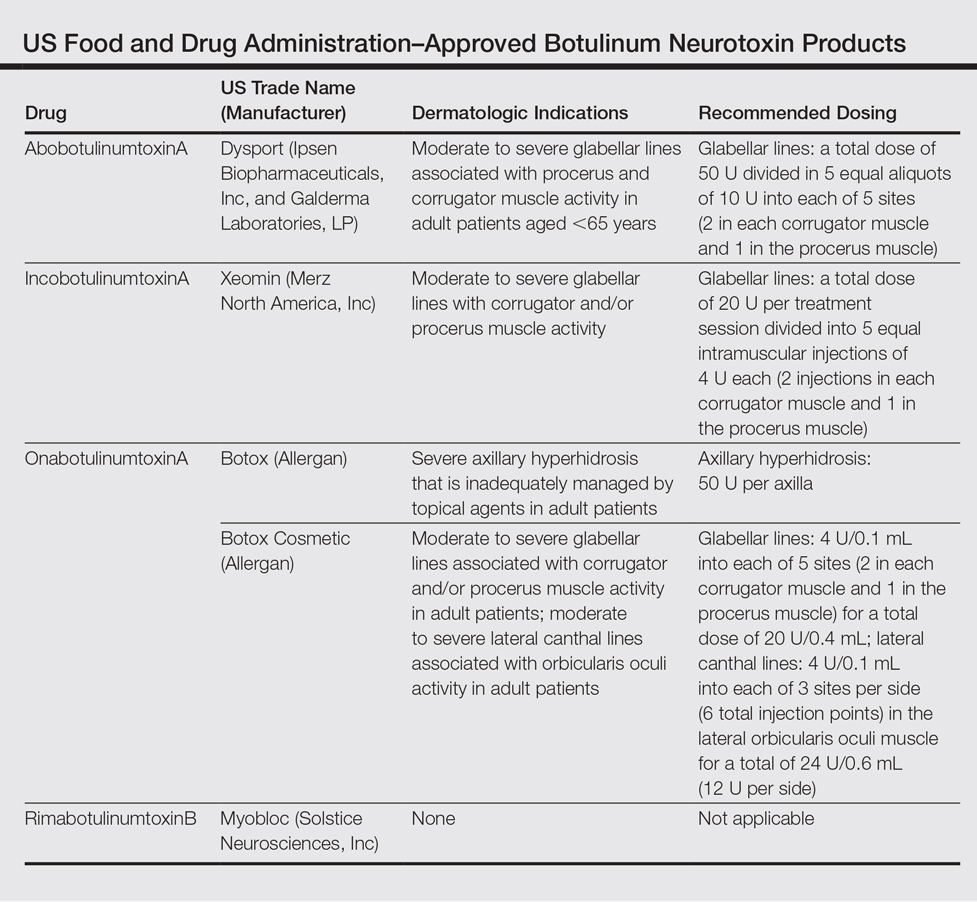
AbbVie : FDA Approves Expanded BOTOX Label for the Treatment of Pediatric Patients with Spasticity | World Pharma Today

ABBVIE: FDA Approves Expanded BOTOX® (onabotulinumtoxinA) Label for the Treatment of Pediatric Patients with Spasticity | FDA Health News